
homework - How to calculate m/e value for the base peak in mass spectroscopy? - Chemistry Stack Exchange

SOLVED: Calculate the sodium chloride equivalent (the E-value) for a substance (use two decimal places) that has a molecular weight (MW) of 136, is a 2 ion electrolyte (and dissociates 80%). The





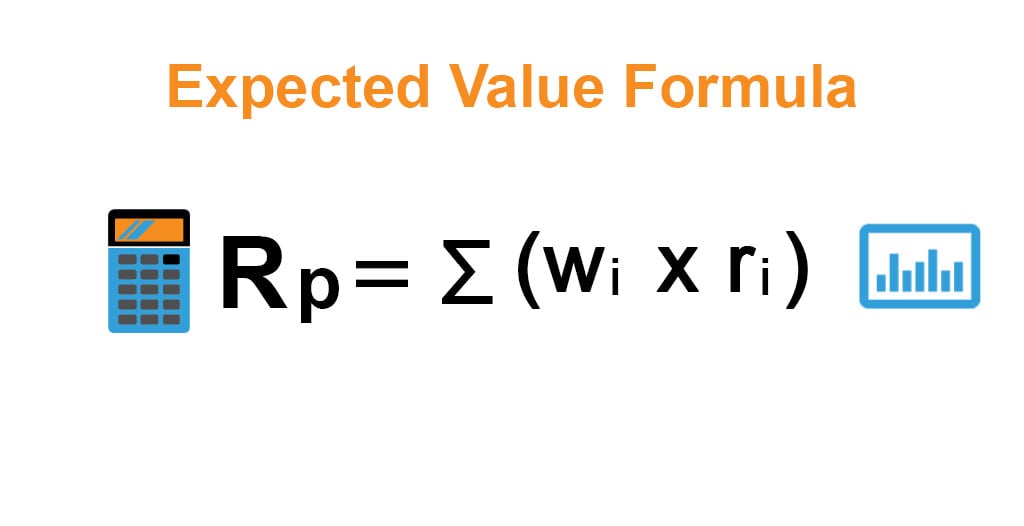
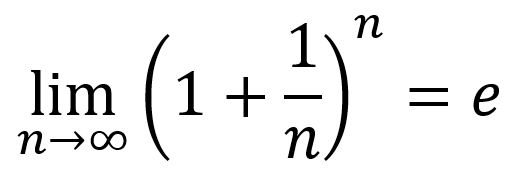


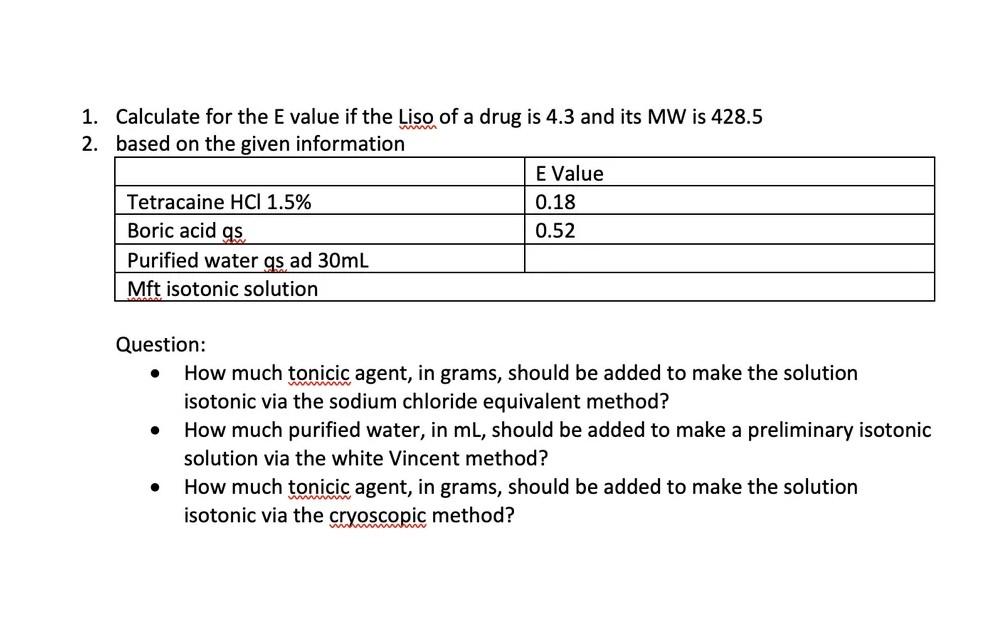
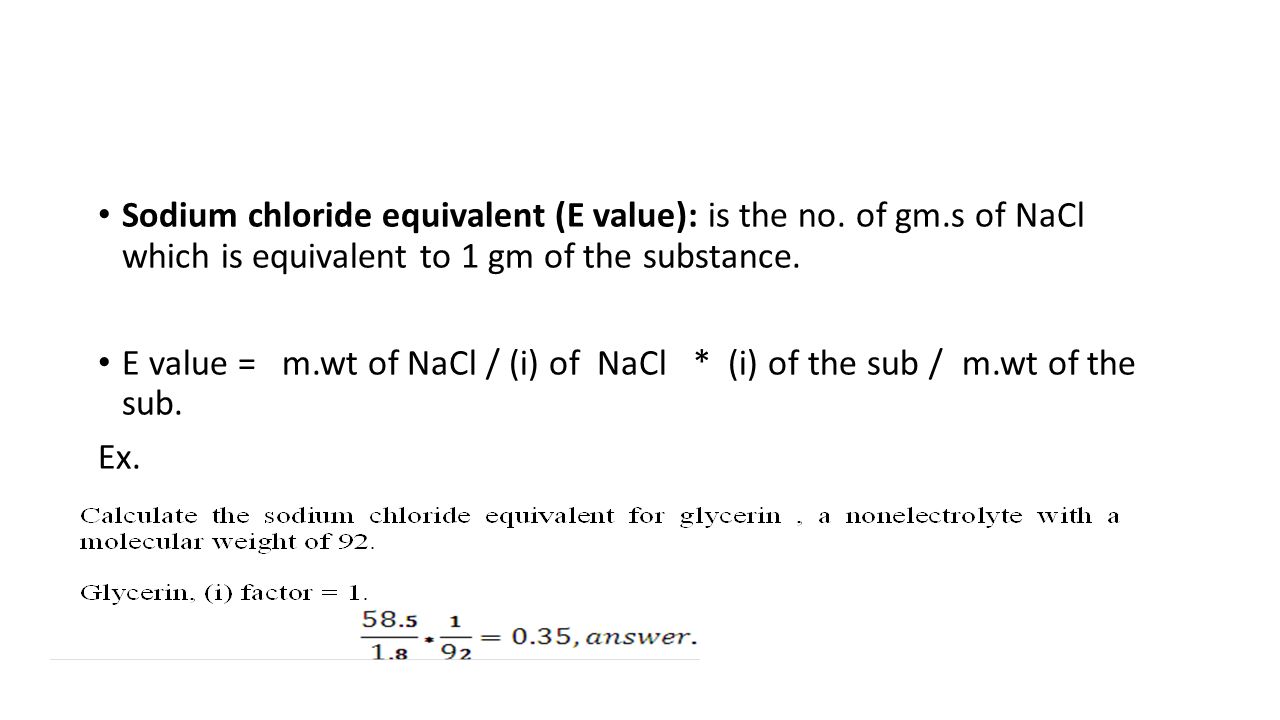

:max_bytes(150000):strip_icc()/FormulaForExpectedValue-58b8980d3df78c353cc32aff.jpg)


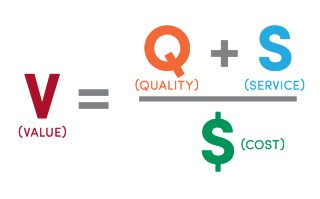

:max_bytes(150000):strip_icc()/Enterprise-value-sales-4197048-FINAL-a98faa4f5e4b40bfa58dd1bd16640386.png)
:max_bytes(150000):strip_icc()/Ending-Market-Value-Final-b17e5c89fe15445986f004c45f80cee4.jpg)
