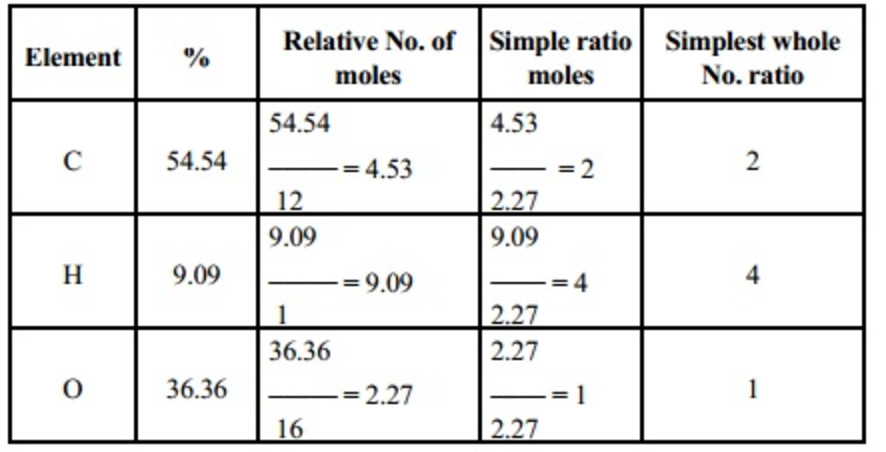
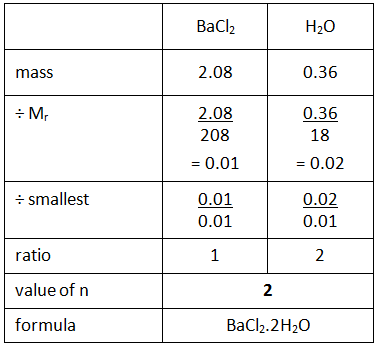
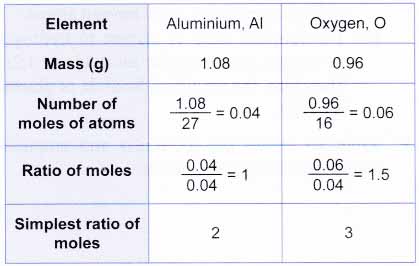
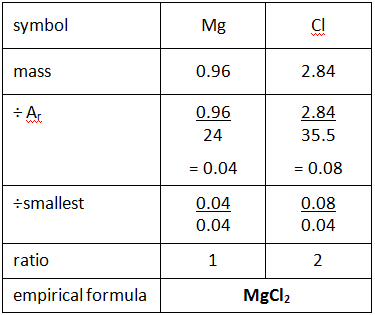
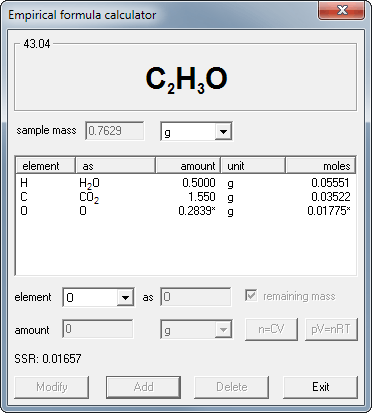
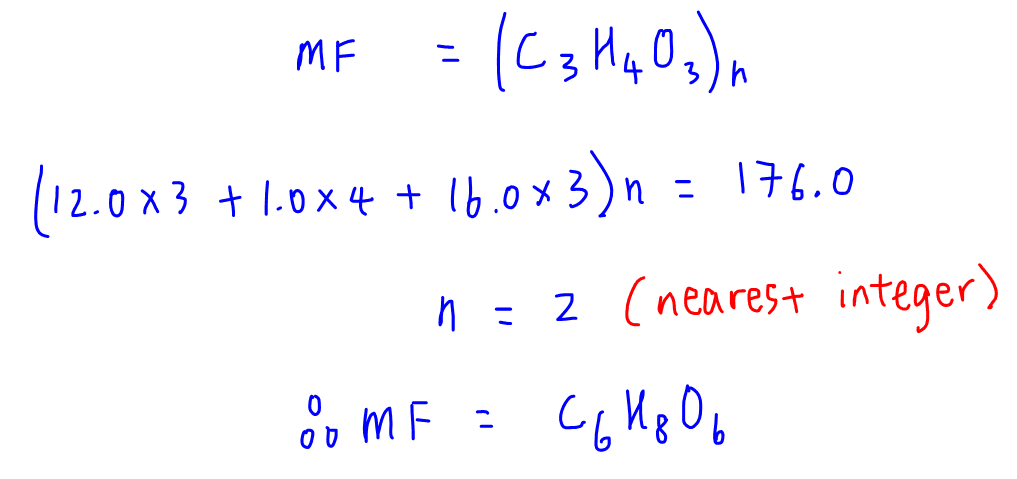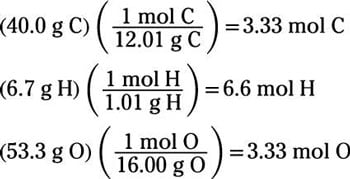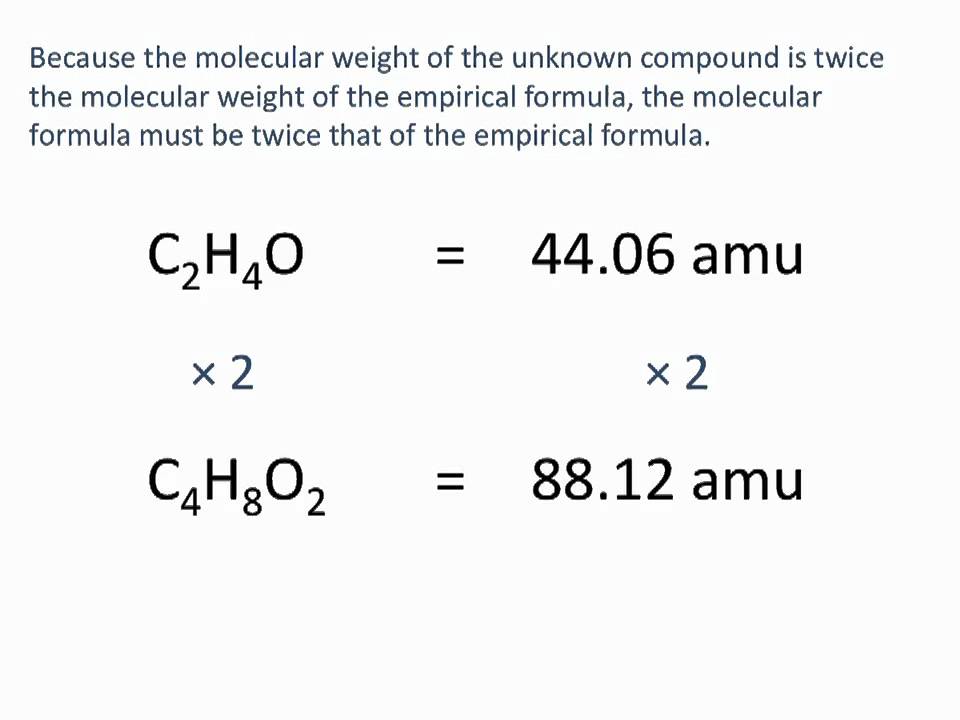Empirical formula and molecular formula. https://youtube.com/@NAJAMACADEMY #chemistry #empirical #empiricalformula #molecular #molecularformula... | By Najam Academy | Calculating molecular formula from empirical formula. In this lecture I will teach ...