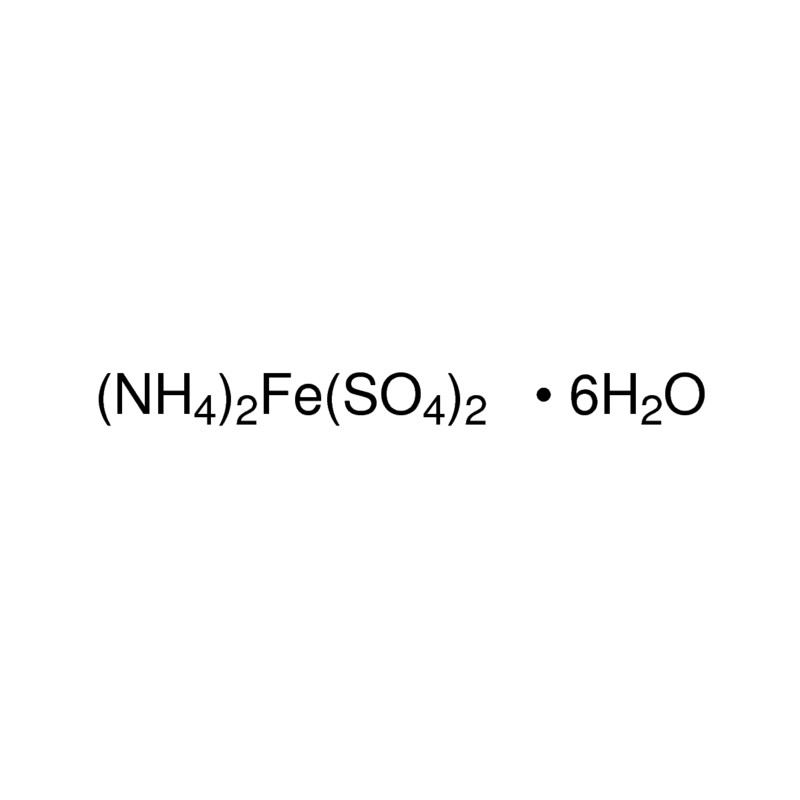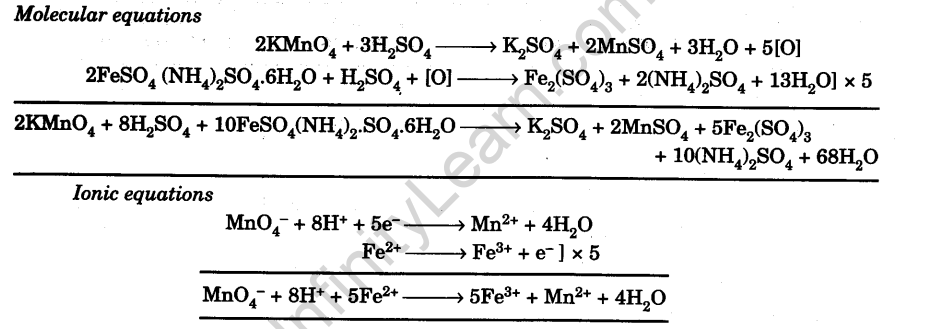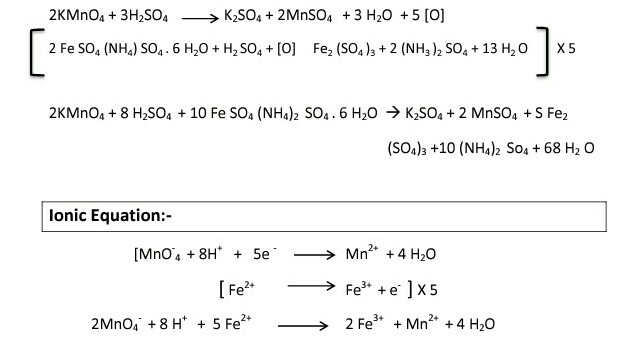
To determine the Molarity of KMnO4 solution by titrating it against a standard solution of Mohr's salt. (M/20 Mohr's salt solution).

Oxidation state of fe in mohr's salt with solution and formula Step wise explanation needed - Chemistry - Redox Reactions - 16519045 | Meritnation.com