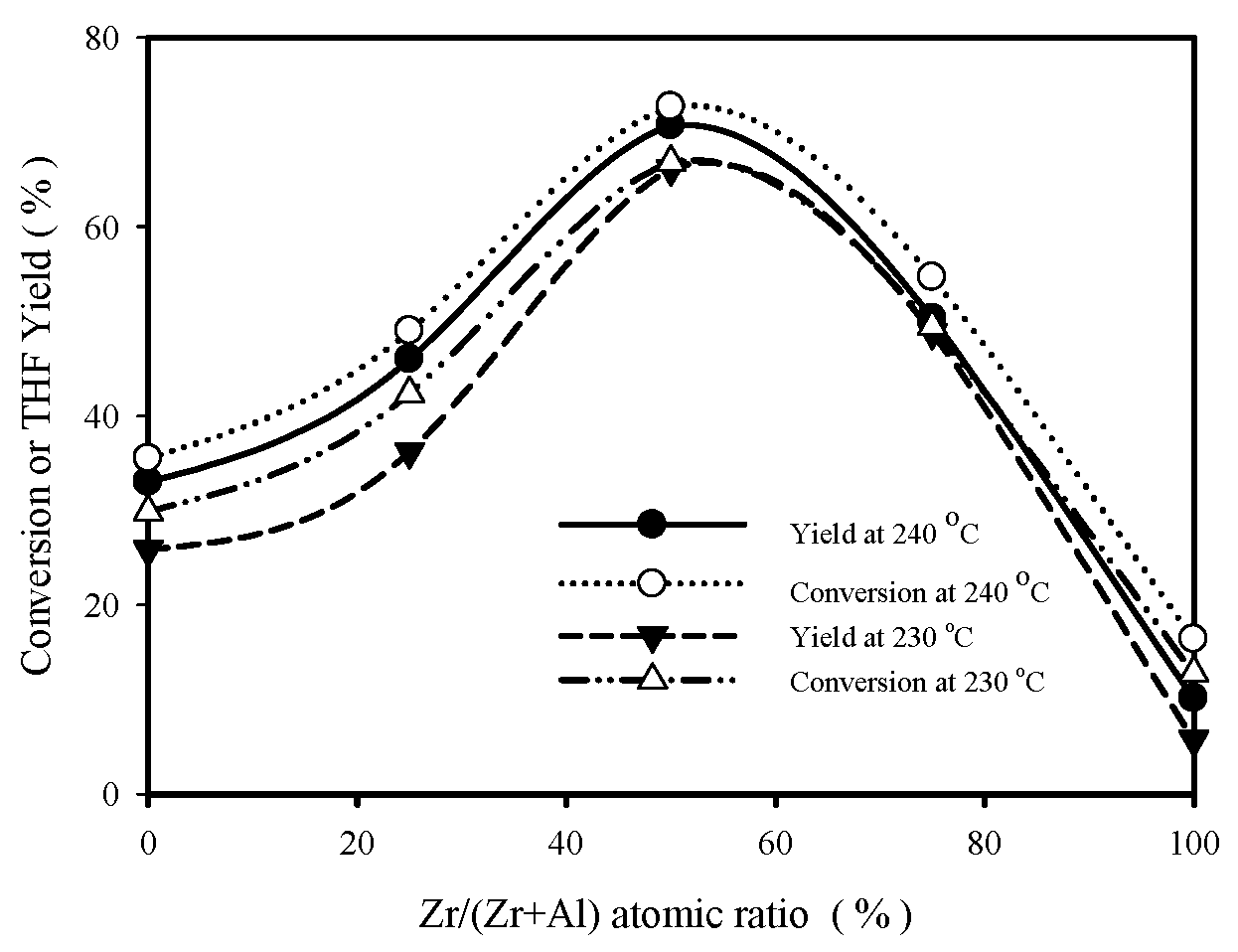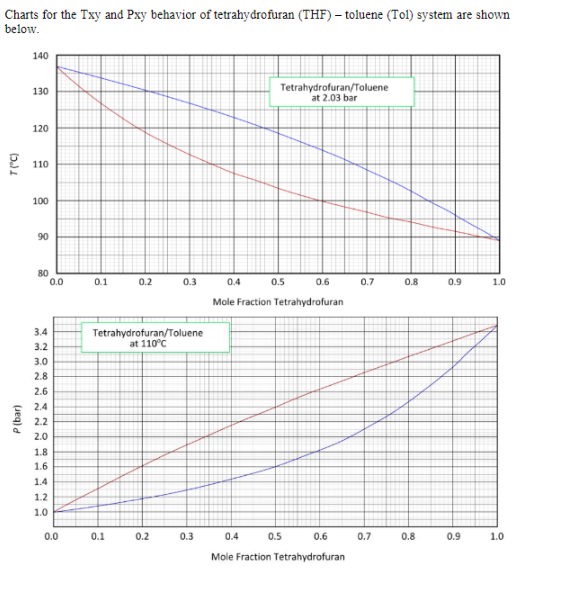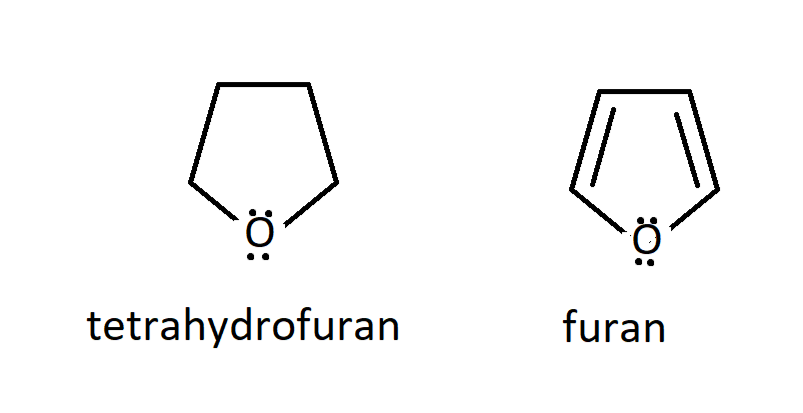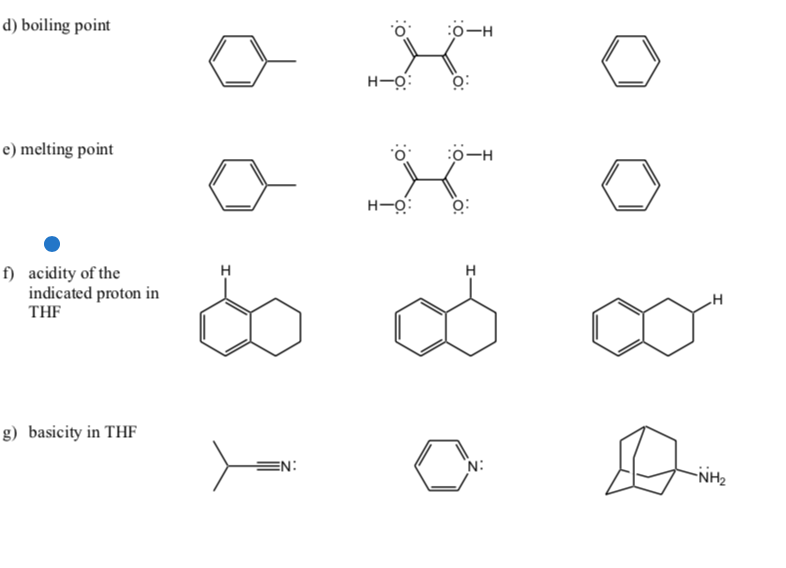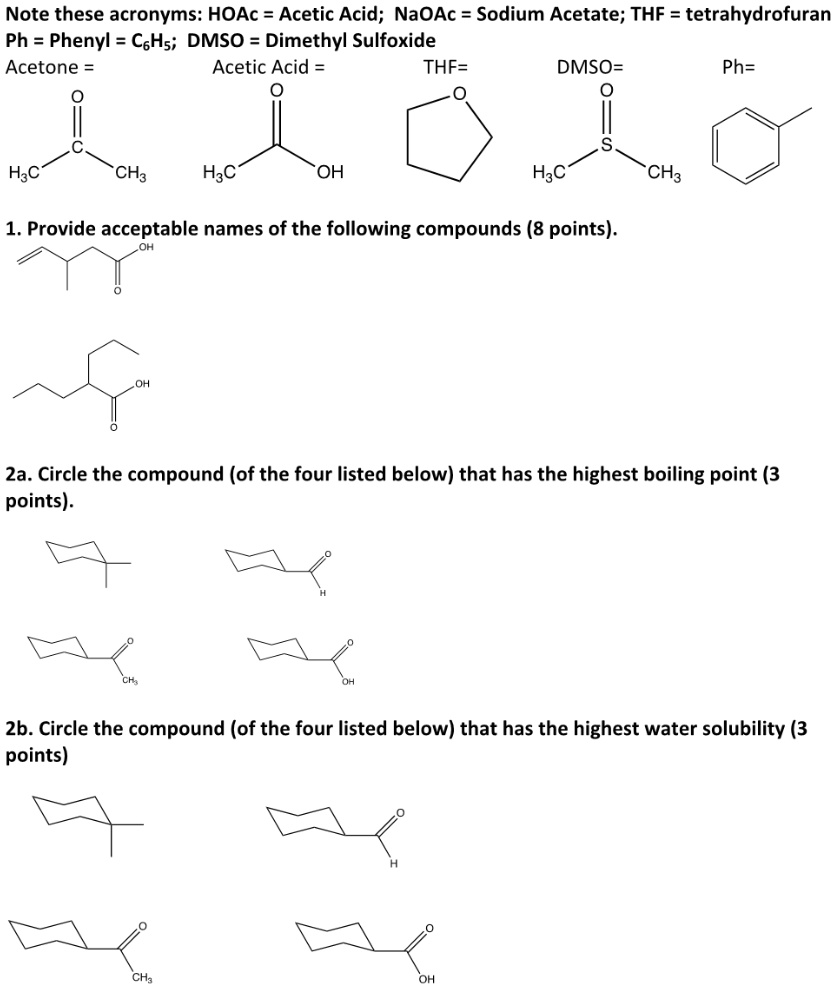
SOLVED: Note these acronyms: HOAc = Acetic Acid; NaOAc = Sodium Acetate; THF = tetrahydrofuran; Ph = Phenyl = C6H5; DMSO = Dimethyl Sulfoxide; Acetone; Acetic Acid; THF; DMSO; Ph. H3C OH
a I0 −1 vs T −1 of the THF-water mixture at various THF contents and b... | Download Scientific Diagram

Thermodynamic Properties of Water + Tetrahydrofuran and Water + 1,4-Dioxane Mixtures at (303.15, 313.15, and 323.15) K | Journal of Chemical & Engineering Data

The effect of pressure on composition and temperature of azeotrope (a)... | Download Scientific Diagram

Explain why tetrahydrofuran has a higher boiling point and is much more water soluble than furan, even though both compounds are cyclic ethers containing four carbons. | Homework.Study.com